Instruction Manual
Table Of Contents
- Glossary of terms
- Assistance
- Information on this manual
- Safety
- Reception
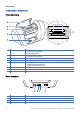
- Product description
- Getting started
- Operating with VR NxT
- Maintenance of the instrument
- Troubleshooting and errors
- Technical features
- ANNEX A: CELLSEARCH sample preparation for DEPArray
- ANNEX B: DEPArray recoveries preparation for downstream genetic analysis
- License Agreement
- Conformity
- Warranty and Responsibility
12 VR NxT| Instruction Manual 1.0 02-2019|VRNXTinstr1EN|© 2019 Menarini Silicon Biosystems SpA
Reception
Storage warnings
NOTICE: Keep the packing materials to move or store the instrument in future. The instrument must be stored indoor in
its original packaging to avoid exposure to adverse weather conditions.
Identification
Manufacturer's contacts
Menarini Silicon Biosystems SpA
Via Giuseppe Di Vittorio 21b/3
40013 Castel Maggiore (BO)
Italia
www.siliconbiosystems.com
Instrument identification label
Part Description
Product description
Part number
Serial number
Manufacturer data with date of manufacture.
Dispose the instrument as electronic waste (European Community Directive 2012/19/EU).
Consult the instruction for use for important cautionary information.
Consult the instruction manual.
CEmark, declaration of conformity for Europe
ULmark, with ULfile number 12.5
FCC ID FCC certification ID number