User's Manual
Table Of Contents
- 1 Using this guide
- 2 Applicable Documents
- 3 About C-Scan®
- 4 Indications, Contraindications, Adverse Events, Warnings, Cautions
- 5 C-Scan® system
- 6 Instruction for use for the clinic team
- 6.1 Preparations before procedure
- 6.2 C-Scan® Cap ingestion procedure
- 6.3 During the procedure days (from C-Scan® Cap ingestion to excretion)
- 6.4 Radioactive source not properly concealed – malfunction notification instruction
- 6.5 Post procedure- after C-Scan® Cap excretion
- 6.6 Analysis & report reception
- 6.7 C-Scan® user interface notifications
- 7 Instruction for use to the subject
- 7.1 Permitted activities during the procedure:
- 7.2 Prohibited activities during the procedure:
- 7.3 C-Scan® procedure
- 7.4 Daily routine during the procedure
- 7.5 Post procedure- after C-Scan® Cap excretion:
- 7.6 When to contact the Clinic
- 7.7 Analysis & report reception
- 7.8 C-Scan® user interface notifications
- 8 C-Scan® View Installation Guide
- 9 Using C-Scan® View Application
- 9.1 C-Scan® View Application Login Screen
- 9.2 C-Scan View Updates
- 9.3 C-Scan® Track Data Download and Upload to Server
- 9.4 Add a Failed Procedure to Database
- 9.5 Managing your local files
- 9.6 C-Scan® View Editor
- 9.6.1 Editor introduction
- 9.6.2 Slice Profile
- 9.6.3 2D XRF/CMT Map
- 9.6.4 Structural Model
- 9.6.5 2D Map Timeline
- 9.6.6 Play Bar
- 9.6.7 General Buttons
- 9.6.8 Report on Case Failure
- 9.6.9 Estimated Colon Path
- 9.6.10 Bookmarks
- 9.6.11 Suspect Metrics
- 9.6.12 Suspect Wizard
- 9.6.13 Pre-analysis Summary
- 9.6.14 Report Preview
- 10 Technical Description
- 11 Troubleshooting- C-Scan® View
Document Title
C-Scan System User Reference Manual
Document No. DOC0001667 Revision 14 Date 22 May 2022
Page 76 of 90
Before using this document, check that you use latest revision (see Arena)
* Printed revisions are for Reference Only*
Figure 92: Procedure Quality
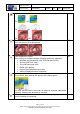
Findings table (#3 in Figure 91)
If suspicious findings were reported during pre-analysis, their information and pictures
appear at the top of the Findings table (see #1 Figure 93).
Figure 93:Findings Table
A suspicious finding is automatically marked to appear in procedure report. A series of non-
suspicious findings appear bellow the SF’s (see #2 Figure 93) if there was any.
Suspect’s information (slice number, time in procedure and location) is displayed in the left
side bellow the suspect’s classification (Suspicious/Non-suspicious finding). Suspect’s
parameters are displayed In the Analysis Findings column, next to the analysis flow.
Browsing snapshots gallery can be done using the right and left arrows, next to the snapshot.
Snapshot number out of total number of snapshots is displayed on the top left corner of the
snapshot gallery.
Snapshots can be deleted using the delete button .
Comments column shows comments entered during suspect analysis process and can be
edited or added.
Non-suspicious findings are not marked to appear in the report but can be manually added
to the report by checking the box near the suspect comments.
All findings can be viewed with additional data directly in analysis window by clicking the
Analysis button (see #5 in Figure 91) or the jump to suspect analysis button to jump
to a specific suspect in the analysis window. Return to pre-analysis summary by clicking the
Show summary button. Going back to analysis window, changes can be made to pre-
analysis; suspects can be added/removed.
Procedure summary, comments and recommendations (see #4 in Figure 91)